Reagent Array Drug Sample Selection and Acquisition Program Uses PharmaKB Content
Key Takeaway
The Pharmaceutical KnowledgeBase (PharmaKB) content may be used within 3rd party commercial applications. In this case, Reagent Array, a specialty screening sample provider, enables drug sample selection based on key properties from the PharmaKB database.
Reagent Array Drug Sample Selection
Reagent Array subscribes to a custom data feed from CDD’s new Pharma KnowledgeBase (PharmaKB) drug profile information on FDA and EMA approved drugs and drugs in clinical trials. Reagent Array employs content from the PharmaKB to allow researchers to select drug ingredient samples.
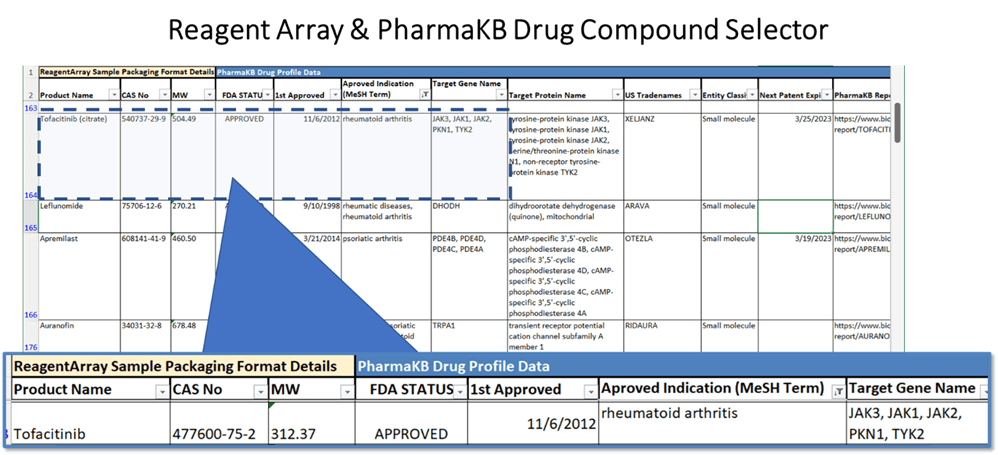
PharmKB data fields used for sample selection:
- Common drug name and trade names.
- Biological target and reference codes.
- FDA 1st approval date & current status.
- FDA Approved disease indications.
- Common Industry identifiers.
- Exclusivity impacting patent end dates.
- Entity type (Small molecule, Protein, etc.)
Reagent Array Sample Packaging
Reagent Array samples are delivered in simple single use cassettes holding 24 samples at 0.5 to 2 microliters each. An example sample cassette is illustrated in Figure 2. The sample cassette system provides for delivery of 24 samples to a standard screening place in one simple step.
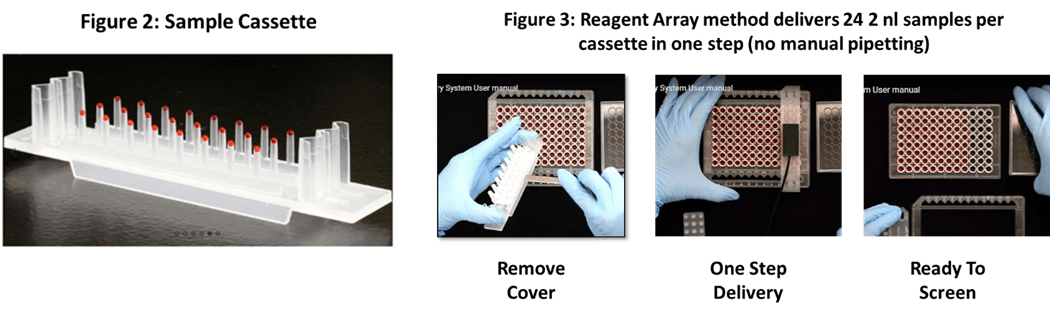
Standard Reagent Array Sample Formatting
Under Reagent Array standard formatting, each 24 pin cassette contains 8 unique compounds packaged at three different concentrations allowing for easy calculation of dose response curves. Each sample cassette is provided in duplicate so results may be validated.

Reagent Array and CDD are working together to provide drug discovery researchers access to user selected custom sets of FDA approved drug ingredients packaged as cost-effective single use laboratory sample sets in screening programs. These custom sets come in a screening-ready format saving both time and money.
For information on Reagent Array products and services:
Please visit: www.ReagentArray.com
Email: info@ReagentArray.com
For information on Collaborative Drug Discovery’s Pharma KnowledgeBase (PharmKB):
All content is available through the PharmaKB web application at (www.pharmakb.com) or via the PharmaKB API (details at https://www.pharmakb.com/api-documentation).
Email: info@pharmakb.com
